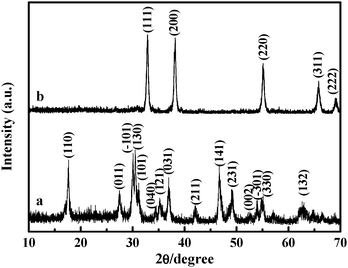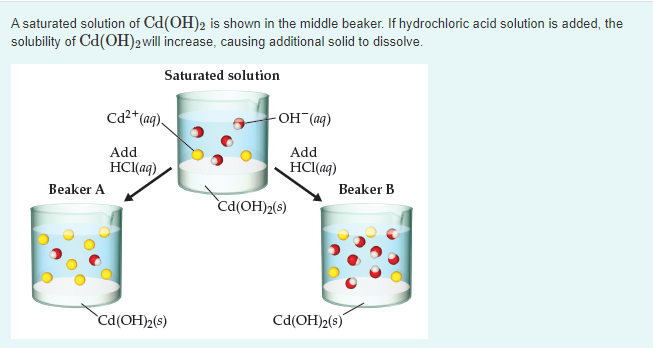
Nanomaterials | Free Full-Text | In Situ Fabrication of CdS/Cd(OH)2 for Effective Visible Light-Driven Photocatalysis
21041-95-2 Cadmium hydroxide, China 21041-95-2 Cadmium hydroxide Manufacturers, China 21041-95-2 Cadmium hydroxide Suppliers - china-reagent

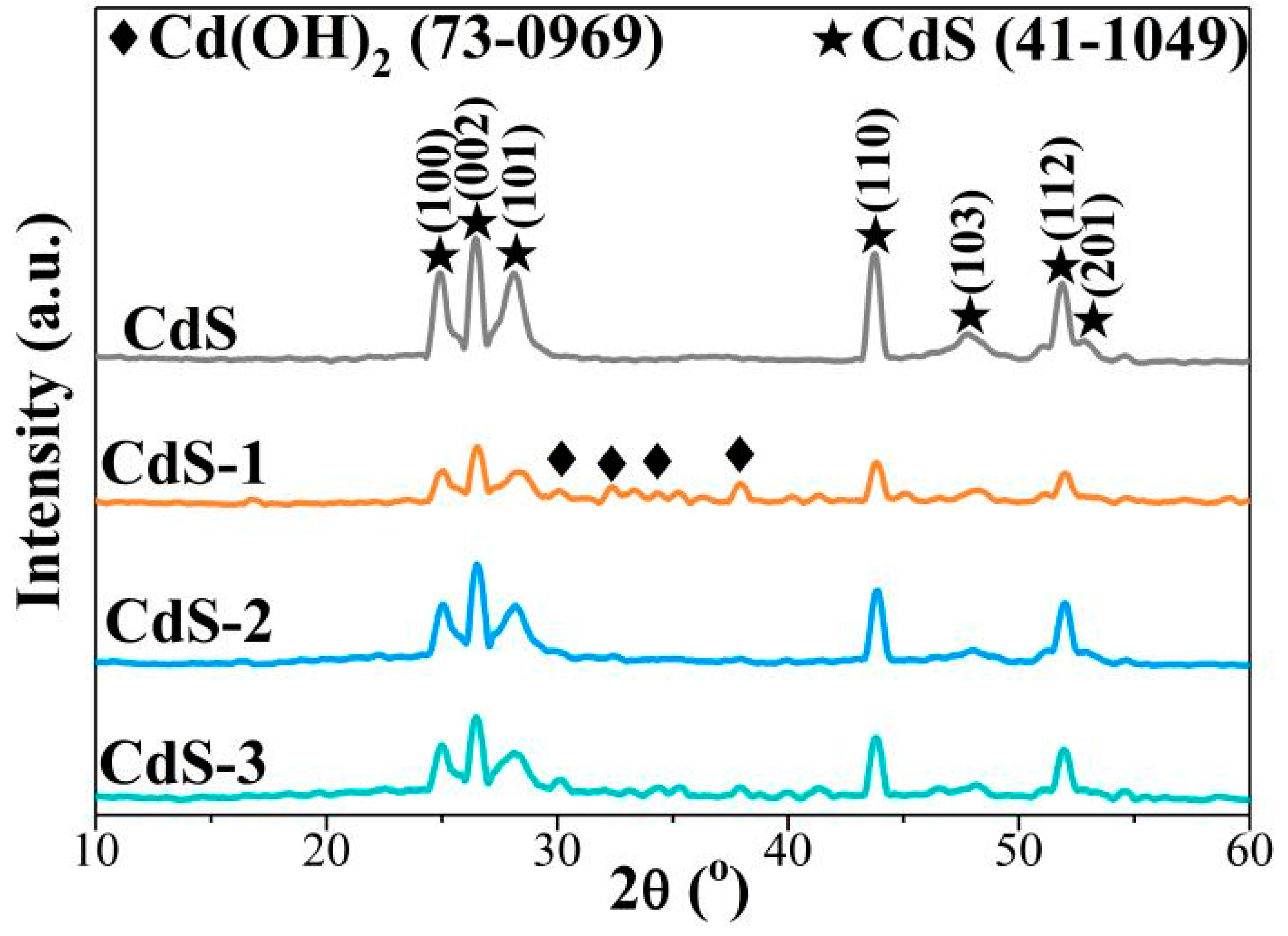
Remarkable positive effect of Cd(OH)2 on CdS semiconductor for visible-light photocatalytic H2 production - ScienceDirect

Remarkable positive effect of Cd(OH)2 on CdS semiconductor for visible-light photocatalytic H2 production - ScienceDirect

SOLVED: What is the pH for a saturated solution of Cd(OH)2? The Ksp for Cd( OH)2 is 5.9 x 10^–15. pH =

Equilibrium d 36. The solubility of Cd(OH), pH = 13 25°C is (Ksp of Cd(OH)2 = 2.5 10-14) (1) 2.7 * 10-13 (2) 1.09 10-10 (3) 2.5 * 10-12 (4) 1.6 10-14
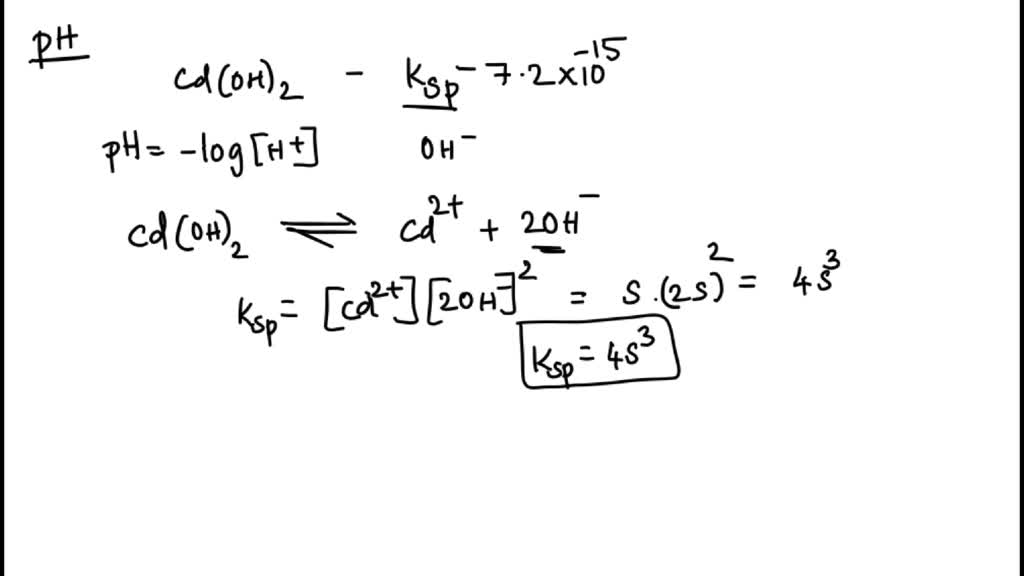
SOLVED: Calculate the pH of a saturated solution of cadmium hydroxide; Cd(OH )2. Report the answer to decimal places. Ksp = 7.2x10^-15.